A new category between external fixation and definitive reconstruction
Temporary Internal Stabilisation for Critical Long-Bone Defects
A modular intramedullary system designed for staged reconstruction, PMMA spacer support, and infection-conscious temporary fixation.
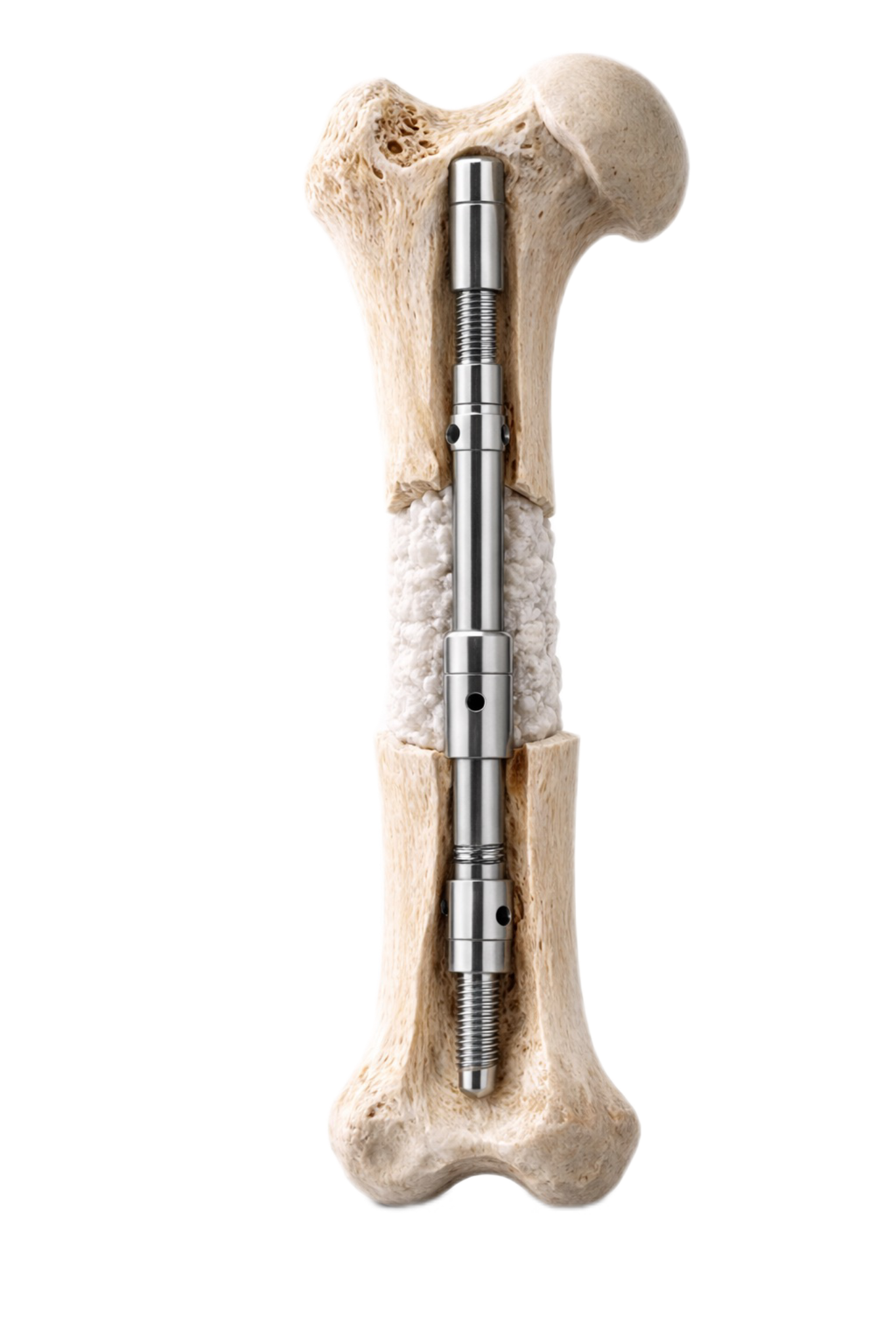
Where CITadel fits in the treatment pathway
Designed for temporary internal stabilisation in staged limb reconstruction.
Current limitations in managing critical bone defects
No dedicated internal solution for staged defect management
External fixation increases complications and patient burden
Standard implants are not designed for spacer-supported constructs
Reconstruction pathways remain mechanically suboptimal
CITadel: a modular temporary internal stabilisation system
Modular architecture
Two-component system adaptable to defect size and anatomy.
Mechanical-axis support
Intramedullary alignment for stable temporary fixation.
PMMA spacer compatibility
Designed to function with antibiotic-loaded spacers.
HA-Ag antibacterial interface
Selective coating in bone-contact zones.
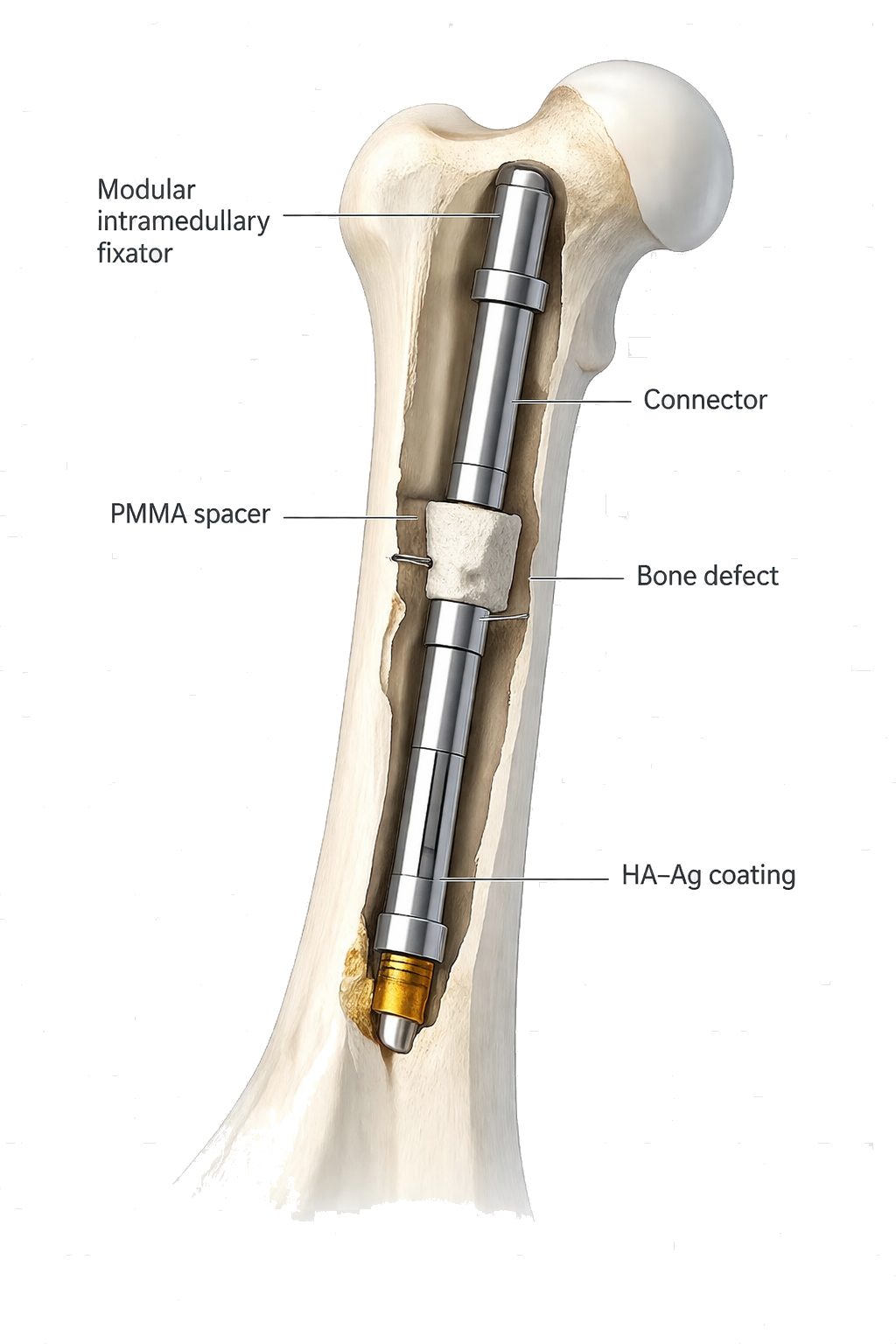
How CITadel is used in staged reconstruction
Debridement
Debridement and defect creation
PMMA Spacer
PMMA spacer placement
CITadel Fixation
Internal fixation with CITadel system
Reconstruction
Definitive bone reconstruction
Designed for staged treatment, not single-stage defect replacement.
Primary clinical use cases
Infected critical bone defects
Osteomyelitis cases requiring staged debridement, antibiotic spacer support, and delayed reconstruction.
Blast and high-energy trauma
Complex limb injuries with segmental bone loss requiring temporary internal stabilisation.
Oncologic segmental resection
Post-resection defects needing stable internal fixation during staged limb salvage.
Why CITadel is different
Current solutions
- External fixation dependency
- Lack of spacer-compatible systems
- Limited internal options for defects
CITadel
- Internal temporary stabilisation
- Designed for spacer-based workflows
- Defect-focused construct logic
Clinical & Economic Impact
Positioned as a cost-saving treatment protocol
Development and validation status
CITadel has undergone biomechanical testing and early clinical use. Further development and pilot implementation are planned with industrial partners.
Current TRL: 4–5 → Target: TRL 6 (clinical pilot)
Partnership and collaboration opportunities
Industrial licensing
License the CITadel system for manufacturing and distribution within your product portfolio.
Co-development
Joint engineering and regulatory pathway development towards CE/MDR compliance.
Clinical pilot sites
Partner as a reference centre for structured clinical validation and pilot deployment.
Strategic investment
Accelerate transition to clinical-scale adoption through strategic funding.
Interested in working with us? Let's start the conversation.
About the inventor

Maksym Baida, PhD
Orthopaedic Trauma Surgeon · Developer of CITadel
Over 15 years of clinical practice in traumatology and reconstructive orthopaedics. Associate Professor at Bogomolets National Medical University, Kyiv. CITadel was born from a direct clinical need — the absence of a reliable internal stabilisation option for patients requiring staged reconstruction.
Contact and inquiries
CITadel is open for collaboration with industry partners, clinical centres, and strategic stakeholders.
Send us a message
Describe your interest — we respond within one business day